Almost always, stationary battery rooms ventilation is an argument among engineers. However, seldom do their reasoning are based on scientific evidence to prove their position. IEC std 62485 provides a solution to the ventilation issues for battery rooms.
Definition of Terms
- Stationary Battery
- A secondary battery which is designed for service in a fixed location and is not habitually moved from place to place during the operating life. It is permanently connected to the DC power supply (fixed installation).
- Secondary Battery
- Two or more secondary cells connected together and used as a source of electrical energy.
- Secondary Cell
- An assembly of electrodes and electrolyte which constitutes the basic unit of a secondary battery.
NOTE This assembly is contained in an individual case and closed by a cover.
Why is Ventilation for Stationary Battery Rooms Required?
During charge, float charge, and overcharge of a battery, gases are emitted from all secondary cells and batteries excluding gastight sealed (secondary) cells. This is a result of the electrolysis of the water by the charging current. Gases produced are hydrogen and oxygen. When emitted into the surrounding atmosphere an explosive mixture may be created if the hydrogen concentration exceeds 4 %vol hydrogen in air.
When a secondary cell reaches its fully charged state water electrolysis occurs according to the Faraday's law. When the operation of the charge equipment is stopped, the emission of gas from the cells can be regarded as having come to an end up to one hour after having switched off the charging current.
Ventilation Requirements
The purpose of ventilating a battery location or enclosure is to maintain the hydrogen concentration below the 4 %vol hydrogen Lower Explosion Limit (LEL) threshold. Battery locations and enclosures are to be considered as safe from explosions, when by natural or forced (artificial) ventilation, the concentration of hydrogen is kept below this safe limit.
The minimum air flow rate for ventilation of a battery location or compartment shall be calculated by the following formula:
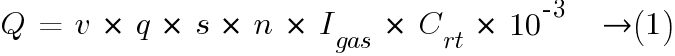
where:
Q = ventilation air flow in m3/h;
v = necessary dilution of hydrogen: 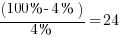 ;
;
q = 0.42 × 10-3 m3/Ah generated hydrogen at 0 °C ;
Remark: for calculations at 25 °C, the value of q at 0 °C shall be multiplied by factor 1.095.
s = 5, general safety factor;
n is the number of cells;
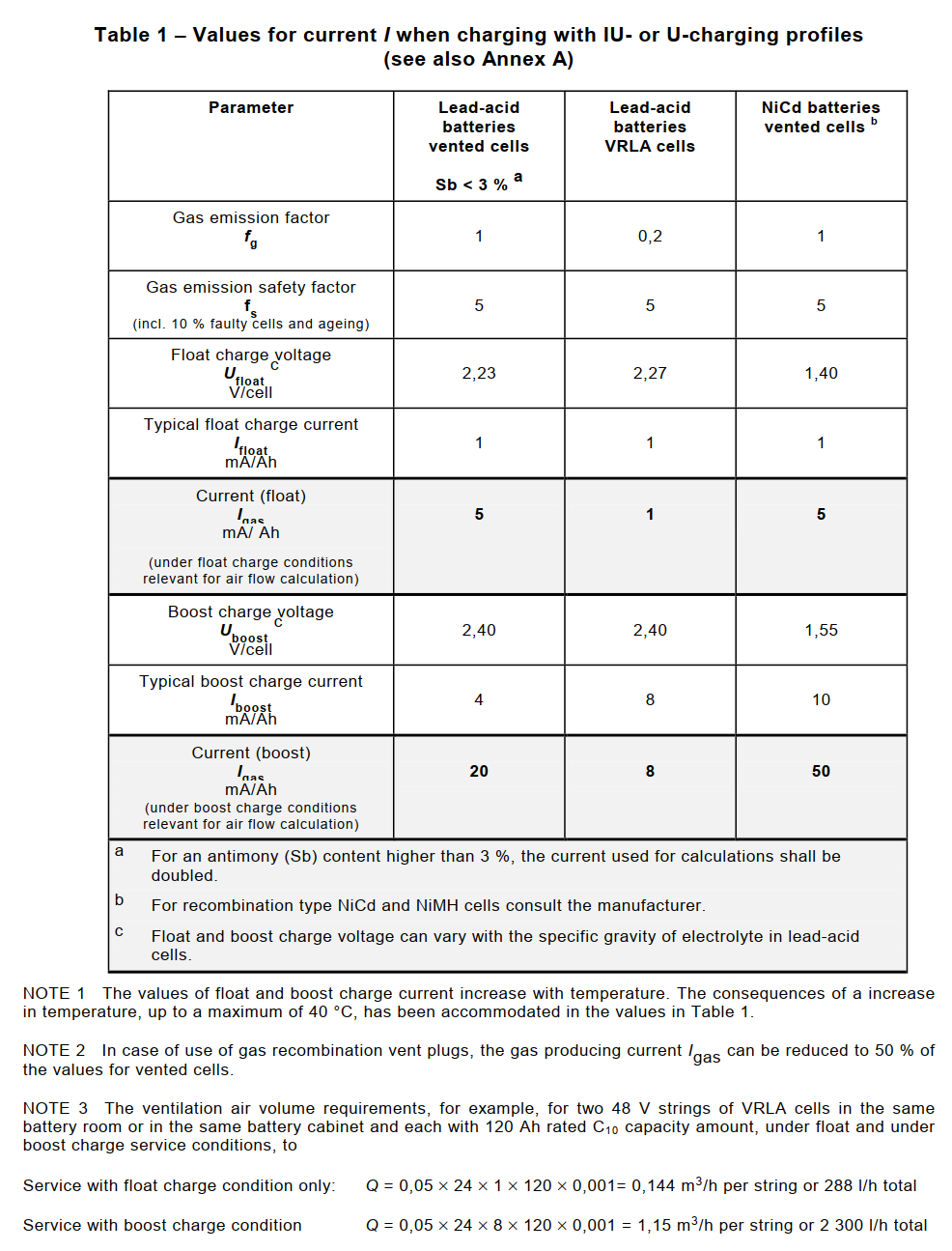
Igas = the current producing gas in mA/Ah rated capacity for the float charge current Ifloat, or the boost charge current Iboost;
Crt is the C10 capacity for lead acid cells (Ah), Uf = 1.80 V/cell at 20 °C or C5 capacity for NiCd cells (Ah), Uf = 1.00 V/cell at 20 °C.
With v × q × s = 0.05 m3/Ah the ventilation air flow calculation formula is:
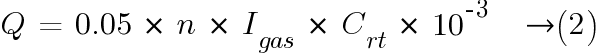
The current Igas producing gas is determined by the following formula:
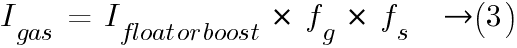
where:
Ifloat = float charge current under fully charged condition at a defined float charge voltage at 20 °C;
Iboost = boost charge current under fully charged condition at a defined boost charge voltage at 20 °C;
fg = gas emission factor, proportion of current at fully charged state producing hydrogen;
fs = safety factor, to accommodate faulty cells in a battery string and an aged battery.
Unless otherwise stated by the manufacturer, the preferred values for Ifloat and Iboost with supporting data are given in the following Table 1.
Source: IEC 62485-2:2010
